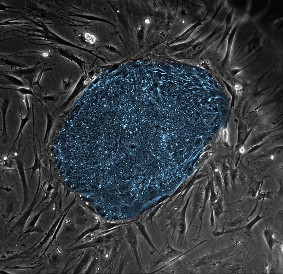
Human embryonic stem cell research is an area of special interest to NIGMS. It represents a unique opportunity to explore the most fundamental mechanisms of biology and development while providing a foundation for future clinical applications. NIGMS’s support of basic research in embryonic stem cell biology is actually greater than that of any other component of NIH.
As part of our continuing commitment to this research area, we hosted our third workshop that brought together 54 NIGMS grantees working on human embryonic stem cells. Each workshop helps them exchange ideas and pursue collaborations while informing us about their progress and challenges.
The research presented during the talks and poster session at this year’s meeting covered a broad array of topics, reflecting the most up-to-date (and unpublished) work from labs across the country. It was wonderful to see how much progress has been made since the previous workshop two years ago! Sessions focused specifically on advances that help us understand how cells self-renew, how and when differentiation occurs and what directs cell fates. Another session discussed technological developments, such as large-scale culture techniques and the application of cutting-edge approaches in proteomics, glycoproteomics and global mapping of chromosomal interactions. Grantees also shared their latest progress on induced pluripotent stem cells and genetic reprogramming.
After listening to the presentations, it became clear to me that one of the pivotal directions for future stem cell research is going to be epigenetics, especially as it relates to regulating pluripotency, directing cell fate and inducing genetic reprogramming. Several talks, for instance, showed how two different but genetically similar human embryonic stem cell lines give rise to distinct cell types under the same conditions, presumably due to pre-existing epigenetic marks.
Since the last meeting in 2007, we’ve seen the remarkable development of human induced pluripotent stem cells. But as Jamie Thomson suggested in the closing session, we won’t be able to decipher critical differences between these cells and human embryonic stem cells until we really understand the range of variability in both types of cells. Given the accelerating pace of progress, I expect that future research will generate many new insights and perhaps some surprises that we’ll hear about in the next two years.