UPDATE: The Maximizing Access to Research Careers (MARC), Undergraduate Research Training Initiative for Student Enhancement (U-RISE), Initiative for Maximizing Student Development (IMSD), Graduate Research Training Initiative for Student Enhancement (G-RISE), Bridges to the Baccalaureate, and Bridges to the Doctorate funding opportunity announcements are now available.
NIGMS has a longstanding commitment to developing a diverse pool of biomedical scientists through a variety of institutional training and student development programs. Based on stakeholders’ feedback through Requests for Information (NOT-GM-15-108; NOT-GM-17-017), as well as extensive analyses and discussions with NIH staff and the community, we intend to make adjustments to our programs designed to enhance the diversity in the biomedical research workforce. The modifications, which the NIGMS Council recently approved, are designed to: 1) provide equity of trainee support across programs; 2) prevent programmatic overlap; 3) align the funding strategies with the programmatic goals; 4) tailor expectation of outcomes, support mechanisms, and review considerations according to the institution’s level of research activity; and 5) strengthen our ability to evaluate the success of the programs. The changes, described in more detail in the recent Videocast of the Council Open Session, will impact the Initiative for Maximizing Student Development (IMSD), the Research Initiative for Scientific Enhancement (RISE) program, and the Maximizing Access to Research Careers – Undergraduate Student Training in Academic Research (MARC U-STAR) programs. We don’t anticipate any immediate changes to our Postbaccalaureate Research Education Program (PREP). Possible adjustments to the Bridges to the Baccalaureate and Bridges to the Doctorate programs are currently under discussion.


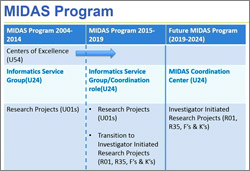


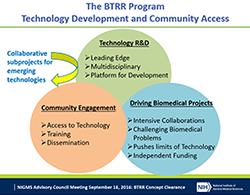

 At last week’s Advisory Council meeting, I presented a report on the comments we received in response to our request for information (RFI) on a potential new program for research funding.
At last week’s Advisory Council meeting, I presented a report on the comments we received in response to our request for information (RFI) on a potential new program for research funding.