Now that we have completed the review process for Maximizing Investigators’ Research Award (MIRA) applications from the first eligible cohort of established investigators, I would like to update you on the program’s status and plans for its future. I shared this information with our Advisory Council at its recent meeting in January.
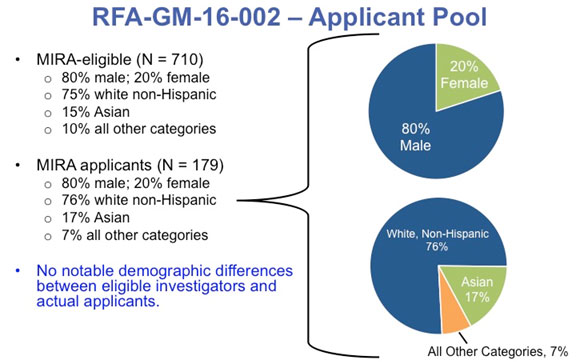
The first funding opportunity announcement (FOA) we issued (RFA-GM-16-002) was for established investigators who had either two NIGMS R01s or one NIGMS R01 for more than $400,000 in direct costs. In either case, one grant had to be expiring in 2016 or 2017. Out of the 710 investigators who could have met these criteria, 179 submitted applications, corresponding to 25% of the eligible pool.
Among the eligible investigators, 80% were male and 20% were female. This ratio was unchanged among those who applied, as were the percentages across racial and ethnic groups (Figure 1). Thus, although the demographics of the group of investigators that was eligible for this first FOA were skewed in several ways, the skewing was not exacerbated in those who chose to apply.
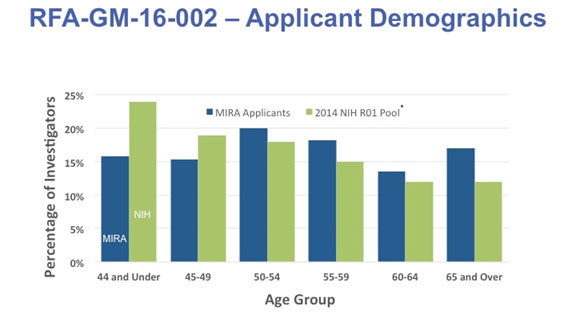
As one might have expected of well-funded, established investigators, the age distribution of the applicants was shifted toward older PIs relative to all researchers who have NIH R01 funding (Figure 2). The age distribution was essentially the same for the eligible and applicant pools (not shown).
The applications were distributed among four review panels managed by the NIGMS Office of Scientific Review. These panels were organized around broad scientific areas and included reviewers with expertise in a range of fields. Prior to the panel meetings, the reviewers participated in extensive orientation webinars designed to familiarize them with the goals of the program, discuss how MIRA grants and review processes differ from those for R01s, and answer any questions or concerns that might have arisen as the panelists began reading applications. We also introduced the panelists to recent data regarding implicit bias and its implications for peer review.
Since MIRA applications did not require specific aims, applicants were instructed to outline a broad vision for the direction their work would take over the next 5 years, with the understanding that they could change directions as new ideas, observations and opportunities arose. As described in the FOA and answers to frequently asked questions, we asked reviewers to focus on the applicant’s scientific track record, the importance of the research questions being addressed and the overall strategy proposed to answer them, while at the same time deemphasizing the details of approach and methodology. Reviewers also considered applicants’ record of service to the research community and contributions to developing the biomedical research workforce.
The results of this first round of review are encouraging. Staff members who observed the meetings reported that the reviewers understood the goals of the program and review process. There were no significant differences in mean priority score as a function of applicants’ age, gender, race or ethnicity. We will continue to monitor the process closely for any evidence of biases as the program expands. We are also exploring ways to measure differences in emphasis in written critiques between MIRA and standard R01 reviews to test our impression that the reviews were substantially more “big picture” than is typically the case in traditional study sections.
Using the information from the study sections and input from our Advisory Council, we are in the process of making funding decisions for this first group of applications.
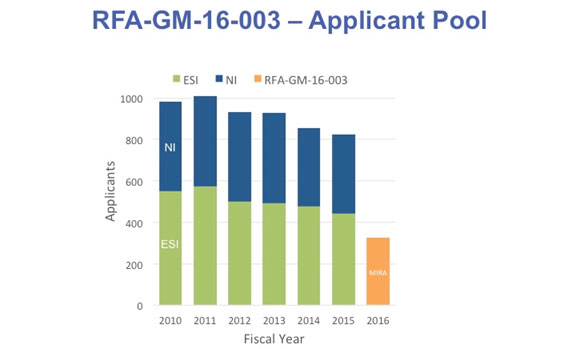
The second part of this phase of the MIRA pilot was an FOA (RFA-GM-16-003) for early stage investigators (ESIs) and new investigators (NIs) at the assistant professor or equivalent level. We received 326 applications from eligible investigators for this FOA. Between 2010 and 2015, the Institute received 800-1000 applications per year from new and early stage investigators (Figure 3). Only a fraction of NIs are eligible to apply for the MIRA FOA, and thus we estimate that 30-50% of the possible pool applied for an NI/ESI MIRA. These applications will be reviewed by the NIH Center for Scientific Review in March.
We plan to modify and reissue the established investigator MIRA FOA so that another set of PIs can apply. We intend to use the same criteria for funded NIGMS grants as in the first FOA, with a shift in the eligibility window to allow applicants who did not have a grant expiring in Fiscal Year 2016 or 2017 to apply this round. In addition, we will modify the FOA language slightly based on what we learned from the first cycle of review.
We will also analyze the results from the ESI/NI MIRA pilot and determine whether to reissue a modified version of this FOA as well.
Finally, we are starting to work on plans to expand the program to allow more NIGMS investigators to apply for MIRA grants. Our goal is to issue an FOA that would let anyone with an NIGMS R01 apply for a MIRA in the review cycle in which he or she would normally have submitted an R01 renewal application. This strategy will allow PIs who are currently supported by NIGMS to decide whether they want to try to keep their R01 or convert it to a MIRA, and it will enable us to manage the number of MIRA applications per review cycle in a rational way. We hope to launch this phase of the program within the next year. Stay tuned!



Thank you for this update. I really hope this funding model takes off and I can join the first cohort.
According to the RFA’s, the original plan was to fund up to 100 EI-mira’s and 70 ESI/NI-mira’s. Is that still the case, or do you have revised numbers on the “UP-TO” level? I’m just trying to predict the success rate, which if these numbers are shooting for the upper bound, would mean an amazing 55% for the EI pool and a harder 21% for the ESI/NI pool. Or do you think there will be an adjustment to these numbers to make the success rates at least equal between the EI and ESI/NI pool?
The numbers in the FOAs were rough estimates. How many grants we award will depend on the number of meritorious applications and the available funds. More applications could be funded than the numbers given in the FOAs.
Will you roll out MIRA for mid-career investigators who have one R01? Or will we need to first earn a 2nd R01 before converting to MIRA?
As stated at the end of the post, our hope is to open the MIRA program to any investigator with an NIGMS R01 in the coming year.
Excellent and timely feedback. In my view, the MIRA funding model is an innovation. It appears to be more “organic” and “flexible” than the traditional RO1 model, and intuitively seems more compatible with the manner in which ideas, hypotheses, and unexpected discoveries emerge from focused and sustained lines of research.
Could we please see the data to support this statement? “There were no significant differences in mean priority score as a function of applicants’ age, gender, race or ethnicity.”
To protect the confidentiality of applicants, NIGMS can’t provide data about review scores for small or moderately sized groups.
In discussing the MIRA with some colleagues, it became clear that many hadn’t heard about. I’d suggest communicating with the eligible pool in addition to the RFA. I was surprised at the low application rate (based on what appear to be advantages to the program)
Thanks for the update. In the new FOA, will investigators currently on a no-cost extension on their NIGMS be eligible?
Since the new FOA has not yet been published, we can’t say with certainty what the eligibility criteria will be this time. But we do expect it to be similar to the previous FOA, except for advancing the time windows. For details, see the MIRA FAQ section on eligibility information and the MIRA eligibility flowchart.
The requirement for the investigator to have received NIGMS grant awards during a particular time period is not affected by a no-cost extension. But the requirement for the investigator to have at least one grant expiring during a particular time period can be affected by a no-cost extension.
How much has it cost administratively to institute the MIRA program?
The implementation of MIRA, as for any new program, has required additional effort by the very dedicated staff at NIGMS and CSR. However, no new personnel were required to accommodate the program. Because applications to the MIRA program replace applications we would otherwise have received for R01s, the increased workload was not due to duplicated effort but rather because of the need to establish new procedures. As the MIRA program expands, we actually expect an increase in administrative efficiency over and above the already efficient process we currently have because of a decrease in the number of applications we receive.
The people I know who received MIRA awards have all had a ~25% budget cut per year based on their current level of NIGMS funding.
When will the funding statistics be available to determine if this is the standard award?
As described in the FOA and the MIRA FAQs, the budget for each awarded application is determined on a case-by-case basis, taking a variety of factors into account, including the level of the PI’s other support and input from study section and our advisory council. We will give further updates on the MIRA program in the coming months.
Thank you for your reply.
Is this true? I was told by my Program Officer that an across-the-board funding system was being used for all awarded MIRA applications. The calculation was the average direct costs in your current RO1’s for the past three years multiplied by 0.85. This essentially means that existing costs cannot be covered and that any new directions that grew out of the work in the last three years and requires additional expenses can’t be covered. Yet, in supporting a MIRA grant, GMS is claiming that: “MIRA provides support for all of the research in an investigator’s laboratory that falls within the mission of NIGMS”. Obviously, this statement of GMS is false, since if one accepts the MIRA award, one may not have the resources to pursue exactly that which may have been proposed in the MIRA application and can’t apply for alternative sources (since GMS claims that it falls within their mission).
So I would warn all investigators, who may have hopes to embark on some new somewhat more costly directions under an existing grant, to stay within the R01 framework and not try to roll that into a MIRA.
A MIRA grant provides support for the NIGMS-related research in a PI’s lab, but will not necessarily pay for the costs of every NIGMS-related research project a PI would like to do. PIs are free to use the funds from a MIRA flexibly, which is one of the benefits of the program, but they will still have to prioritize among the many possible directions they might go with their research. When it comes time to renew the grant, there will be no mention of what was previously proposed – and no previous specific aims to report – and the review will once again focus on what the PI accomplished and the overall direction s/he thinks the work will go in over the next five years. Reviewers will also know the PI’s MIRA budget and can gauge for themselves whether they think the work accomplished was in line with the amount of resources the PI received. For these reasons, the notion that the funds are insufficient for the work proposed does not have the same potential weight as it might for a project-based grant such as an R01.
In terms of other sources of funding, again, because MIRAs do not have specific aims or fund specific projects, there is no proscription against a PI obtaining additional funding from sources other than NIGMS to support his/her research. However, as with all NIGMS research grants, when MIRA funding decisions are made and budgets are set, a PI’s other funding is taken into consideration.
Far from being able to fund an investigator’s full program, the reality of these awards is that they are more like “Minimizing Investigators’ Research Award”. Since the first round was focused on GM’s most successful investigators (those with at least 2 RO1s) the radical cuts will either force them to dismantle their highly successful research programs or force them to leave GM and go elsewhere for their funding. Is GM now much less focused on funding based on scientific merit?
After speaking with several people who have received an offer of a MIRA award, everyone has had a major cut in direct costs. Unfortunately the reduced budgets do not take the cost of conducting the proposed research into serious consideration. Instead, it seems that the primary metric that NIGMS is using to calculate budgets “on a case-by-case basis” is how much other support the investigator receives. Panel scores or the actual cost of getting the work done are secondary considerations, at best. This makes no sense, since non-overlapping research endeavors are not relevant to conducting MIRA-supported research. As a consequence, contrary to MIRA’s goal of reducing the amount of effort a researcher spends writing grants, the budgets will force investigators to spend more time seeking alternative sources of funding. Further, NIH rules make this very challenging because any new proposals an investigator writes will have to avoid the appearance of overlap with the MIRA-funded project.
In line with the last comment, I am currently now in the process of figuring out how to cut 2 salaries out of my lab, along with cutting back on all our animal work (which was the core of the proposal and part of our work over the past 5 years). MIRA clearly equates to Maximizing Investigator’s Retirement Aptitude. It will allow a perhaps leisurely completion of in-progress manuscripts, clearing out the freezers and fridges, sweeping up the floors, and closing the doors. I do now intend to submit another grant that expect to go to another institute, and it is possible that some of the work mentioned in the MIRA application, but which we won’t be able to do might be appropriate for consideration in another institute. But in line with the comments above, the issue of overlap may doom such attempts.
The formula that seems to have applied at my institution for multiple MIRA recipients is 0.88 times the average of the last two years of DCs with no consideration of whether the directions taken in the past 5 years may now justifiably require some modest additional support. Given that the last two years of DCs reflect an already emaciated budget, the additional 12% reduction is not inconsequential. It’s not like taking 12% off of an approved requested itemized budget.
The award will certainly be sufficient to allow a lab (and my lab) to continue to exist, but in a markedly reduced capacity, essentially the equivalent of a generous single non-modular R01. I will learn to see that as a good thing. However, that certainly wasn’t my expectation going into this, and that is probably my fault for not reading between the lines. I do think that GMS should have been more explicit about how the budgets were going to be decided prior to soliciting applications. Nowhere was there an indication that specific budget proposals would not be considered. It was true that the program implied that a budget should try to stay within some range of the existing GMS support, with an upper ceiling given for any individual MIRA, but the idea that specific budget requests and study section recommendations would be ignored was never mentioned to my knowledge. At least, NINCDS in their implementation of a MIRA program allows investigators to also simultaneously submit their competitive R01’s.
Just to make everyone aware of a new wrinkle, MIRA awards are not currently eligible for a diversity supplement. My budget is considerably less that the last 3 years average x 0.85, and ALL of the funding for my lab is through GM. In my case, “other funding” was clearly not a consideration. Perhaps a more apt acronym for MIRA would be “Minimizing Investigator’s Research Award.” Whether I accept the MIRA or not, grant writing seems is in my future.
Similar to other NIGMS research grantees, MIRA PIs will be eligible to apply for supplements, including Research Supplements to Promote Diversity in Health-Related Research. However, NIH is currently in the process of reissuing all FOAs that are affected by the change to new forms, including the FOAs for the various supplement programs. The FOAs will be reissued in stages, with the FOAs for research awards (R01, R03, R15, R21) coming out first, followed by the supplement FOAs.
Until the new supplement FOA is published, the grantee institution may submit a request for a supplement to a MIRA directly to NIGMS following the instructions for paper-based submissions in the existing program announcement. At this time, applications submitted electronically through Grants.gov or eRA Commons will not be accepted for diversity supplement applications to MIRA awards. Please see the NIGMS Diversity Supplements Program Philosophy and/or the Frequently Asked Questions and Answers webpages before submitting an application. Upon publication of the new diversity supplement FOA, an identical application should be submitted as the official application of record, using either paper or electronic submission (note that these use different forms). Applicants should contact the NIGMS diversity supplement program coordinator, Dr. Mercedes Rubio, before applying.
We expect that review of diversity supplement applications will be complete by the time the diversity supplement FOA is published in the NIH Guide and, if the application is approved, an award will be made expeditiously.
It’s really unfortunate that you have to make such a difficult decision. Your work is really terrific and it would be a loss to NIGMS and the research community more broadly if it was not adequately supported.
“I do think that GMS should have been more explicit about how the budgets were going to be decided prior to soliciting applications. ”
This and the other recent comments are very helpful. At our institution funding cuts average 30% of current support and despite what was said on the FOA, there was no negotiation to ensure a smooth transition to deal with the very large and completely unexpected funding cuts.
Thanks for the clarification – I was originally told a MIRA was ineligible.
From the comments here and from MIRA applicants I have spoken with, it is clear that the MIRA program is not accomplishing its stated mission. Recipients must now apply for other funding at the expense of other activities like service or mentorship and will not have much flexibility to explore new areas. In this first round, some recipients did not even get the 5 year benefit because they were required to relinquish current funding before it would normally have terminated. In addition, the awards seem to favor HHMI investigators whose salary does not come from the MIRA award, so fewer positions are cut from their awards than is true for other investigators whose award has a significant salary component. (That said, it unfortunately remains that some post-docs and techs in HHMI labs like non-HHMI labs are vulnerable to losing their jobs due to the unanticipated reduced funding.) Despite these negative consequences, because competing renewals were not allowed and because the MIRA award offers came with the warning that it is unlikely that NIGMS will continue to fund two R01s, most awardees felt compelled to accept the award. So how is NIGMS planning on tracking the impact the MIRA award will have on the first “class” of “awardees”?
The issue of salary support is a very important one. NIGMS enforces the 750K cap regardless of how much institutional support an investigator receives. If this doesn’t change, carrying out NIGMS-funded work at soft money institutions will become close to impossible.
NIGMS reminds investigators that the legislated maximum salary recoverable for 100% professional effort on an NIH research grant is $185,100, excluding fringe benefits. Given other investigator obligations, research effort will generally be less than 100% of professional effort.
So if an investigator needs to cover 80% of his/her salary (plus fringe benefits) from grants, that’s more than 150K. That, alone, is more than half of a typical RO1 and a large chunk of a MIRA budget, especially after significant cuts. Not much $ left to pay postdocs/GRAs and buy supplies.